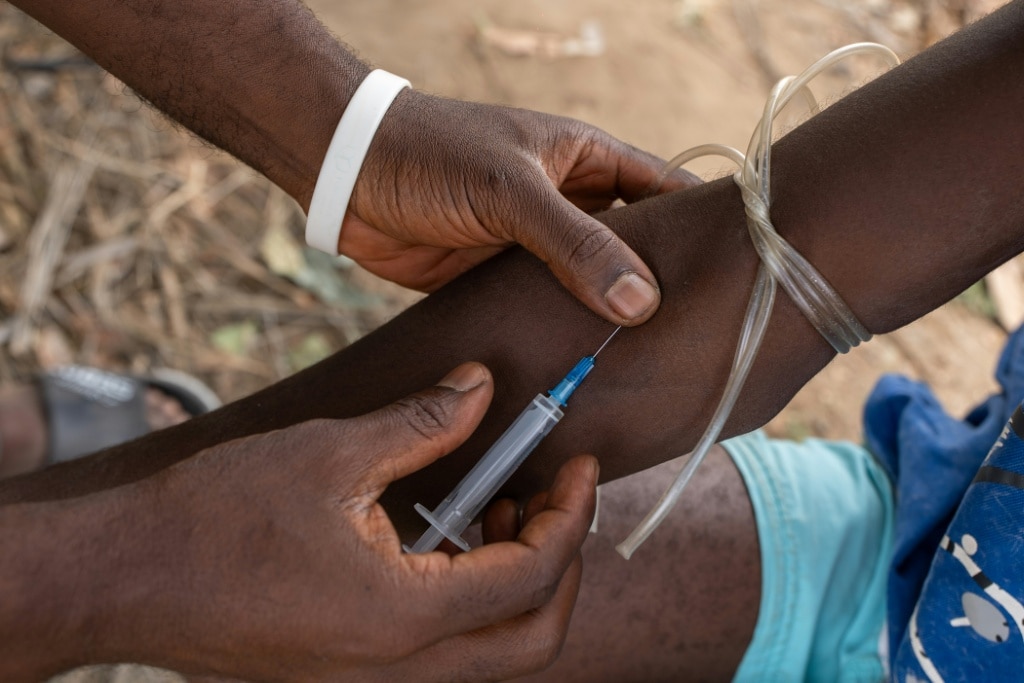
West Africa is currently grappling with an explosive, deadly opioid epidemic that can be traced directly to unregulated, high-strength tapentadol exports from Indian pharmaceutical manufacturers, a months-long Agence France-Presse investigation has confirmed. Sold openly at roadside kiosks and unlicensed street pharmacies across the region, these cheap, unapproved pills are not only driving widespread addiction and death but are also being mixed into the devastating “zombie drug” kush, worsening an already catastrophic public health emergency.
Unlike regulated prescription painkillers sold globally, the tapentadol flooding West African markets comes in doses so potent that no national regulatory authority anywhere in the world has authorized them for human use. Despite India’s 2025 pledge to crack down on this illicit trade after international outcry over harm caused to African communities, customs and shipment records reviewed by AFP show millions of dollars worth of these unapproved high-strength pills continue to flow out of Indian ports every month, bound for Nigeria, Sierra Leone, Ghana and other West African nations where even low doses of tapentadol are explicitly banned. Many shipments are deliberately mislabeled as “Harmless Medicines for Human Consumption” to evade border inspections.
Public health and law enforcement officials across the region say the crisis has reached catastrophic levels. In Sierra Leone, where kush mixed with tapentadol has already been declared a national emergency, authorities collect hundreds of bodies of overdose victims from streets, open-air markets and overcrowded slums every three months — just in the capital Freetown alone. Ansu Konneh, director of mental health at Sierra Leone’s ministry of social welfare, described the addition of tapentadol to the already destructive kush cocktail as “very alarming”. Freetown-based public health researcher Ronald Abu Bangura confirmed that tapentadol is now ground into powder and mixed with kush for distribution, adding that the drug is “being misused all over the place”.
AFP’s investigation cross-checked licensing numbers on seized tapentadol tablets across four West African nations against Indian export records, linking multiple prominent Indian pharmaceutical firms directly to the illicit trade. Gujarat Pharmaceuticals, whose manufacturing license number appeared on seized tablets in both Sierra Leone and Guinea, has been listed in export monitoring databases as a major tapentadol supplier to the region. Merit Organics, another Gujarat-based company, also had its license number found on seized shipments in Guinea. Madhya Pradesh-based McW Healthcare exported more than $1 million worth of 250mg tapentadol tablets to Sierra Leone and Nigeria after India’s February 2025 ban, while PRG Pharma shipped multiple consignments mislabeled as harmless medicines. The investigation also identified Syncom Formulations as the largest single tapentadol exporter to West Africa by value, moving nearly $15 million worth of unapproved pills into the region after the ban, most mislabeled as general harmless medicines.
When contacted by AFP, the Nigerian importer for McW Healthcare was listed at a Lagos address that turned out to be a small camera repair shop with no valid pharmaceutical import permit, with Nigerian health authorities labeling all such shipments explicitly illegal. Notably, PRG Pharma’s director is a shareholder in Maiden Pharmaceuticals, the Indian company blamed by Gambian authorities for the deaths of 69 children in 2023 from contaminated cough syrup.
Experts say the shift of Indian generic drug manufacturers to flood African markets with unapproved opioids follows decades of heavy regulation of opioid sales in wealthy nations, which have seen more than one million opioid-related deaths in the United States alone. For many low-income workers across West Africa, tapentadol is not initially used for recreational purposes: motorbike taxi drivers, market porters and artisanal gold miners take the drug to endure long hours of brutal, back-breaking labor, using it as a makeshift pain reliever and performance enhancer. Abubakar Sesay, a motorbike rider in Freetown who navigates bone-rattling unpaved backroads for a meager income, told AFP, “It energises my body to ride day and night. Without it, I can’t survive.” The pills are also used as an appetite suppressant for people who cannot afford regular meals, and even as a form of currency to pay ransoms for kidnapping victims. Criminal groups and extremist organizations including Boko Haram have also been documented using the drug to build courage for violent attacks.
Nigeria’s National Drug Law Enforcement Agency reports that opioids are now the second most commonly used illicit drug in the country, after cannabis. The agency seized more than two billion high-strength tapentadol pills in 2023 and 2024 alone. Vanda Felbab-Brown, a senior fellow at the Brookings Institution and a leading expert on transnational opioid trafficking, told AFP that weak regulation and limited enforcement capacity in West African nations has created a permissive environment for unscrupulous Indian manufacturers. “This creates opportunities for unscrupulous Indian companies to sell products that are problematic, dangerous, harmful or outright illegal to African countries,” she said. “It’s a prime situation for trafficking networks from India to try to get people hooked.”
A recent report from the Global Initiative Against Transnational Organized Crime found that 90% of all global tramadol seizures over the past decade have occurred in West and Central Africa. After India classified tramadol as a controlled narcotic in 2018, manufacturers simply shifted to producing and exporting tapentadol, which experts note is two to three times more potent than tramadol and far more dangerous. Today, lab testing in Sierra Leone shows that nearly all pills sold on the street as tramadol are actually unregulated high-strength tapentadol, with the similar-sounding name helping the drug slip under the radar of underfunded regulators.
Analysis of shipment data conducted by AFP confirms that nearly 75% of all tapentadol exported to West Africa since India’s 2025 crackdown is the unapproved 225mg and 250mg dosage form. Andrew Somogyi, a professor of pharmacology at the University of Adelaide, told AFP he is not aware of any country that has approved 225mg tapentadol tablets for medical use. He questioned “why a country would want that strength except to bypass regulatory and commercial restrictions”.
India’s national drug regulator, the Central Drugs Standard Control Organisation, told AFP it has “no record” of issuing export approvals for 225mg and 250mg tapentadol, and did not respond to repeated follow-up questions about the ongoing trade. The Indian Drug Manufacturers’ Association, the country’s leading pharmaceutical industry body, defended its members, arguing that legitimate manufacturers who follow domestic export procedures cannot be held responsible for misuse or diversion after the product leaves India. Jaydip Patel of Gujarat Pharmaceuticals told AFP the company’s exports were legal because the importer provided the required authorization letter, and added that manufacturers shifted from tramadol to tapentadol because tapentadol “is easier to export because it is not classified as a narcotic”. When AFP visited Gujarat Pharmaceuticals’ facility in Godhra in January, the building was deserted, with charred tablet fragments and ash scattered across the grounds following a recent fire. None of the other named manufacturers responded to requests for comment.
Across the region, regulatory officials have confirmed that tapentadol of any strength is illegal in their countries. Ghana’s Food and Drugs Authority says it has never issued a permit for tapentadol importation or manufacturing, while Nigeria’s food and drug agency states that any tapentadol found in the country is unauthorised and illegal. Sierra Leone only allows 50mg tramadol to be administered in licensed health facilities, meaning all tapentadol traded on the street is explicitly banned.
Most alarmingly, officials report the drug is now spreading to children and young adults, including primary school students who split tablets into smaller pieces to mix with energy drinks for a stronger high. In Sierra Leone, where the population still carries physical and mental scars from a decade-long civil war, informal detox centers chain addicts to beds for months to force them through cold turkey withdrawal, as the country’s limited official rehab facilities are overwhelmed. Mental health director Konneh noted that many addicts do not even recognize tapentadol as a dangerous drug, because it is packaged and sold as a legitimate medication. “The tragedy is that even addicts seeking help tell us, ‘I’ve stopped taking kush, I’m just taking tapentadol tablets.’ They don’t see that to be a problem to their health,” he said.