Shanghai-based Neuracle Technology has achieved a groundbreaking milestone in medical technology as its implantable brain-machine interface (BMI) system received full market authorization, becoming the world’s first invasive BMI medical device to gain regulatory approval. This historic development signals the transition of China’s invasive neural technology from experimental research to widespread clinical implementation.
The National Medical Products Administration granted the medical device registration certificate on March 13, 2026, following successful clinical trials demonstrating significant improvement in hand-grasping ability among participants with quadriplegia. The device specifically targets patients aged 18 to 60 who have lost grasping function due to cervical spinal cord injuries.
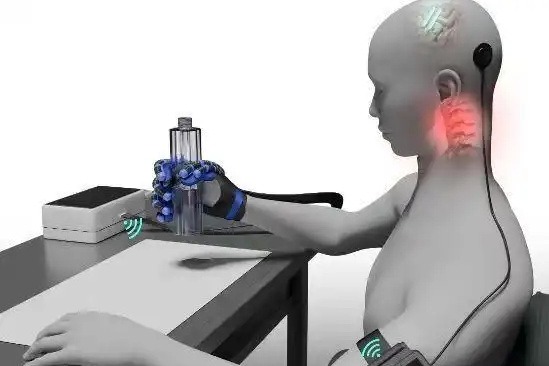
Neuracle’s innovative system employs a coin-sized minimally invasive implant that utilizes extradural implantation technology, positioning electrodes outside the dura mater to avoid brain tissue damage while maintaining high signal quality. The device extracts and analyzes neural signals in real-time, enabling patients to control specialized gloves through thought alone to perform essential tasks like grasping objects and drinking.
Clinical trials involving 36 participants revealed a remarkable 100 percent improvement rate in grasping function, with some patients exhibiting additional neurological benefits including neural remodeling and recovery of supplementary nerve functions. The system features wireless power and communication capabilities, allowing for single implantation with long-term usability. Surgical implantation requires just over an hour, with patients typically achieving independent home operation within one month post-procedure.
Compared to international counterparts, Neuracle’s technology demonstrates superior advantages in trauma reduction, patient compliance, and long-term stability. The company, founded in 2011 and among China’s first BMI-focused enterprises, plans to achieve the first clinical application this year while working to reduce manufacturing costs to enhance accessibility.
The National Medical Products Administration emphasized its commitment to supporting major innovations in high-end medical devices, stating: ‘We will fully promote the application of new technologies, new materials, new processes and new methods in the healthcare field to enhance the international competitiveness of China’s high-end medical devices.’
Industry experts recognize the device as providing a Chinese solution for global neurological treatment and offering novel insights for nervous system rehabilitation. BMI technology represents the convergence of integrated circuits, biomedicine, and artificial intelligence, with Shanghai emerging as a global hub for this cutting-edge field. The city currently hosts approximately 60 BMI companies, representing one of the world’s highest concentrations of firms and most advanced technologies in neural interface development.
By 2025, Shanghai had already seen three invasive products enter China’s special review process for innovative medical devices, with another invasive product entering the FDA’s breakthrough therapy pathway. Several non-invasive BMI products are already deployed in stroke rehabilitation, mental health screening, and sleep disorder treatment.